Alzheimer’s and Autism – Two Sides of the Same Coin

At first glance, autism and Alzheimer’s disease appear to exist at opposite ends of the neurological spectrum, one a condition of early brain development, the other a disease of later-life degeneration. But as our understanding of brain biology deepens, a compelling unifying idea emerges: these two conditions may, in fact, share a common underlying mechanism. That mechanism centers on the regulation of synapses, the vital connections between neurons, and how those connections are shaped, maintained, or lost over time.
We can think of both autism and Alzheimer’s as disorders of synaptic regulation, synaptopathies, a term often used by my colleague, Dr. Dale Bredesen. In the developing brain, synapses are formed in abundance. This is followed by a critical process known as synaptic pruning, during which the brain selectively eliminates weaker or unnecessary connections in a process that refines and optimized neural circuits. In autism, evidence suggests that this pruning process is insufficient. The result is an overabundance of synaptic connections, a state of hyerconnection, like a garden left untrimmed, leading to altered network function and, ultimately, the behavioral and cognitive features we associate with the condition. This situation is now described in attention deficit hyperactivity disorder (ADHD) as well.
Alzheimer’s disease, in many ways, represents the opposite problem. Here, we see excessive synaptic pruning, but occurring later in life and in a pathological context. Some have described this situation as an inappropriate reactivation of the normal developmental synaptic pruning mechanism. Synapses are lost at an accelerated rate, leading to the progressive breakdown of neural networks that underlies memory loss and cognitive decline.
So what connects these two seemingly opposite processes? Increasingly, the answer appears to lie in two fundamental domains: brain energy metabolism and microglial function.
Microglia are the brain’s resident immune cells, but to think of them simply as defenders would be to miss their broader role. They are, in many ways, the brain’s gardeners, constantly surveying the neural landscape, deciding which synapses to preserve and which to eliminate. Their activity is exquisitely sensitive to the brain’s metabolic state. When energy production is efficient and mitochondrial function is intact, microglia tend to maintain a balanced, supportive role. But when energy metabolism falters, due to factors like oxidative stress, inflammation from any source, insulin resistance, or mitochondrial dysfunction, their behavior can shift from being supportive to being destructive. And this shift, and more importantly, preventing it is both the leading edge in neurodegenerative research as well as the central theme of my new book, Brain Defenders.
In autism, altered metabolic signaling during development may impair microglial pruning activity, resulting in the persistence of excess synapses. In Alzheimer’s disease, chronic metabolic stress appears to push microglia into an overactive, inflammatory state, driving excessive synaptic elimination. In both cases, it is not simply the synapses themselves that are the primary issue, but rather the upstream forces that regulate the cells responsible for managing them as demonstrated below:
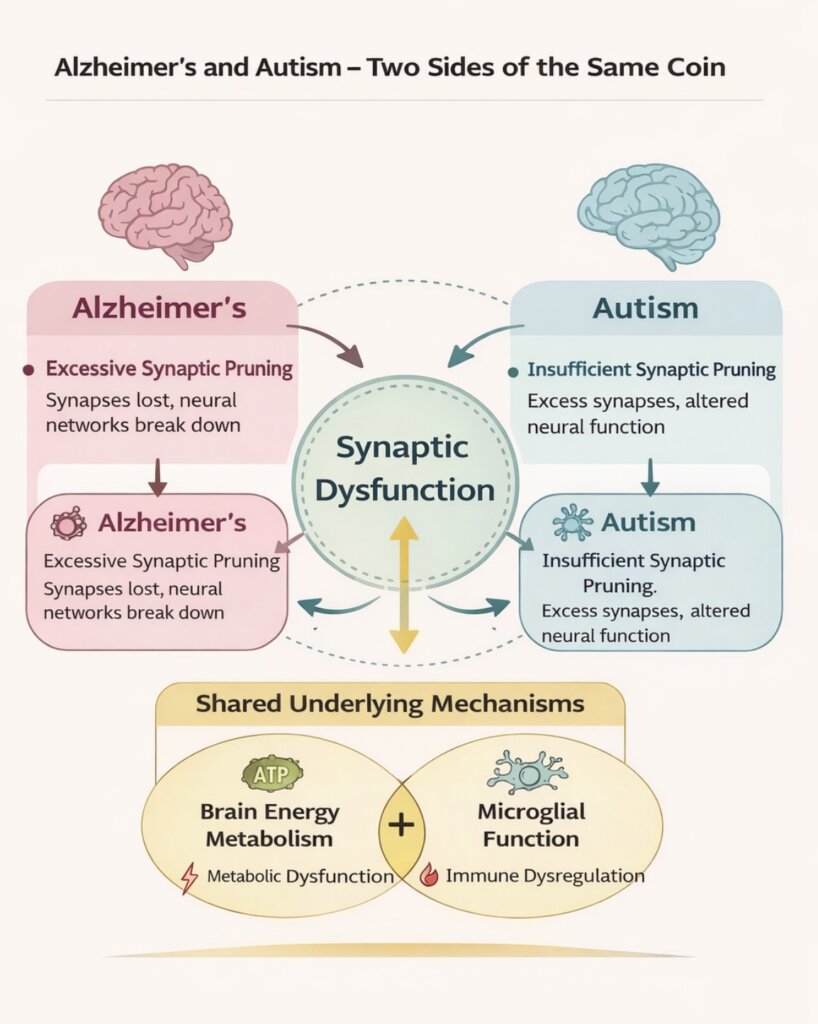
This is where the concept of the brain as an energy-dependent organ becomes central. The brain consumes an extraordinary amount of energy, and even subtle disruptions in energy production can have profound effects on cellular function. When ATP production declines, when oxidative stress rises, and when inflammation takes hold, microglia respond, and in doing so, they shape the fate of synapses.
What emerges is a unifying framework: autism and Alzheimer’s disease may represent two ends of a spectrum of dysregulated synaptic pruning, both rooted in disturbances of brain energetics and immune signaling. This perspective moves us beyond viewing these conditions as isolated entities and instead invites us to consider the deeper biology that connects them.
And importantly, it opens the door to new possibilities. If energy metabolism and microglial function are central to these processes, then interventions aimed at improving mitochondrial health, reducing inflammation, and optimizing metabolic resilience may hold promise, not just for one condition, but across the spectrum of brain health.












